
Stem Cell Research & Therapy
From embryo to stem cell cutting-egde technology

Vietnam, with a population of over 100 million and a rapidly economic development, is emerging as a vibrant location in the stem cell market in Asia.
International Standardisation
SABC's R&D program on stem cell technology and applications is implemented through a collaborative network including reputable R&D structures and legally recognized hospitals and clinics, focusing on three main activities that have a significant impact on the safe and effective clinical therapy and public healthcare: implementing an international standardization, developing a stem cell bank and related products, and updating and transferring advanced stem cell technology.
SABC serves as the foundation for developing CGT technology platform infrastructure in the high-tech zone – a gateway for the transfer of international GMP-cGMP standard technology.
SABC Biobank
Technology Transfer
R&D Therapy Network HLP
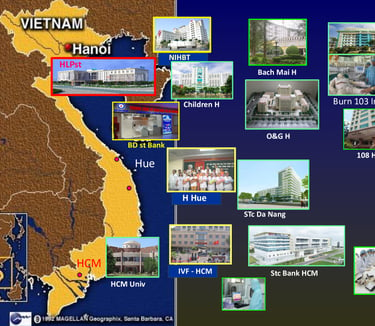
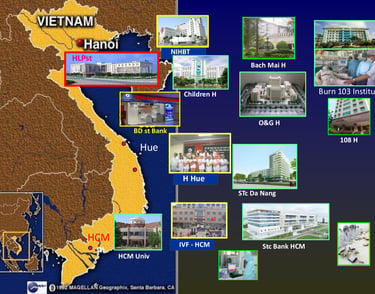
SABC serves as a gateway contributing to the transfer of modern regenerative medicine technologies to reputable treatment organizations in Vietnam such as Pediatric Hospital, Central Obstetrics and Gynecology Hospital, Hanoi Obstetrics Hospital, Cancer Institute, 108 Military Central Hospital, National Institute of Hematology and Blood Transfusion, etc...
SABC has served as president of the Asian Reproductive Biology Society, chairing international conferences and training seminars on reproductive & stem cell technology, cryobiology and regional biodiversity conservation.
SABC Biobank functions as a repository for biological materials, serving as an important asset for connecting with international biobank programs in biomedical R&D, personalized medicine development, and the drug discovery sector. With its unique characteristic of operating in an area with 54 different ethnic minorities, SABC Bank will actively contribute to preserving the world’s human biodiversity and genetic resources, overcoming the lack of diversity in biomedical research.
Vietnam and Southeast Asia are characterized by the expansion of stem cell therapies and open policies regarding application demand, making them strong potential to become a hub for clinical trials and contract manufacturing of cell-based therapies. However, they are limited in investment in basic research, manufacturing, and core technology development compared to other regions. SABC implements a bridging strategy, filling the gap between demand and technology by transferring international cutting-edge advancements in the field of stem cells and gene technology.
Stem cell transplantation has been used effectively as a combination therapy in the treatment of blood cancer and blood disorders in Vietnam. The reality of treatment also reveals challenges to the effective implementation of these technological innovations, such as limited sources of immunocompatible stem cells and the number of cells required to maintain the treatment roadmap. SABC's Biobank program contributes to the proactive production of these replacement cell sources by combining stem cell preservation at -196 degrees Celsius and their expansion culture, including mesenchymal stem cells and induced pluripotent stem cells.



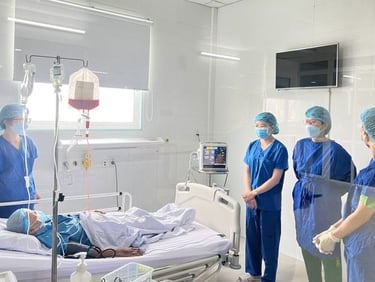
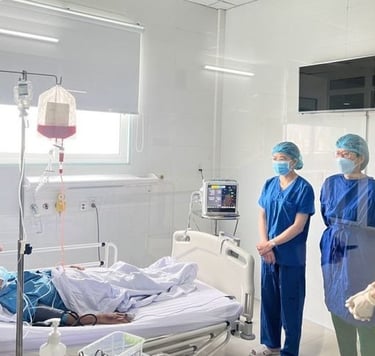

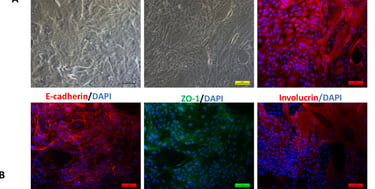
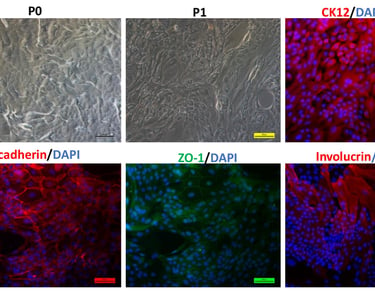




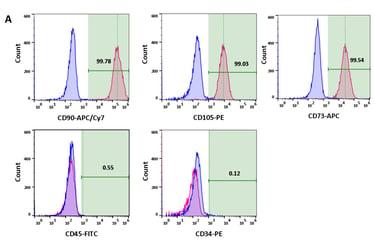










Ø >> Within the framework of state regulations, clinical research and therapy applications have been carried out in many hospitals, demonstrating encouraging positive results. Additionally, the growing market for stem cell applications in therapeutic tourism, healthcare, and aesthetics is creating challenges related to cell quality and products of unproven and illegal origin.

